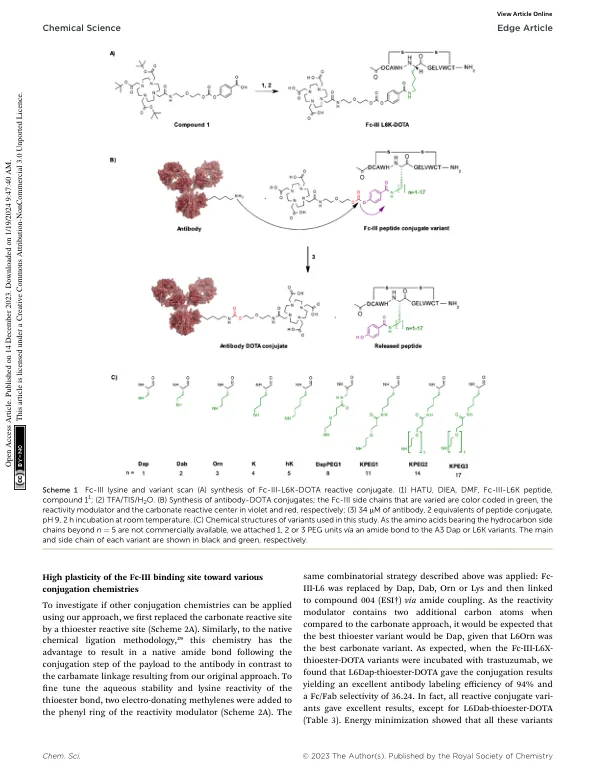
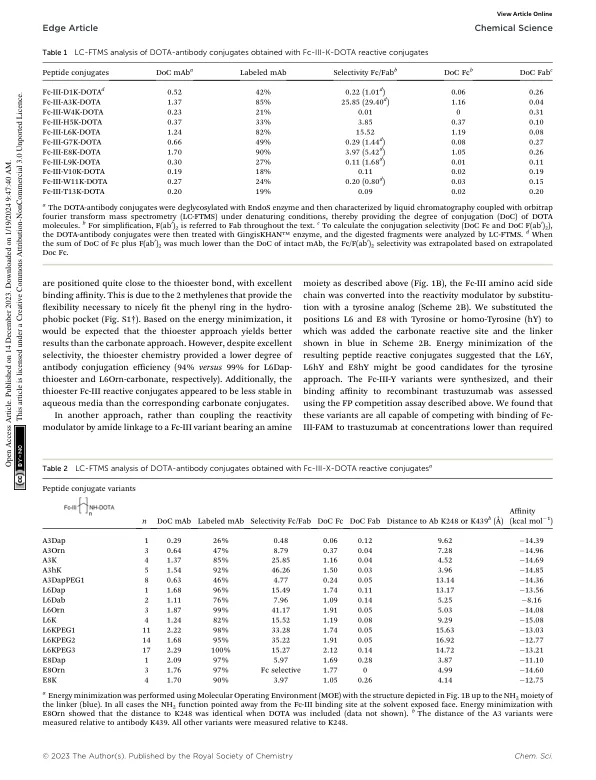
Site-selective template-directed synthesis of antibody Fc conjugates with concomitant ligand release
window of the product, as illustrated by several studies. 2 – 12 In the ADC eld, site-speci c technologies of all types now domi- nate new ADCs entering into clinical trials. However, the recent work of ImmunoGen comparing homogenous and heteroge- nous ADCs that generate the same metabolites, suggests that site-speci c technologies may not always enhance the phar- macokinetics of the drug and may also detrimentally alter its toxicity pro le. 13 – 15 In fact, several criteria such as the nature of the payload, the linker, the conjugation chemistry, the drug- antibody ratio (DAR), the hydrophobicity of the ADC may have an impact on the in vivo properties of the conjugate, which are for the time being di ffi cult to predict. The large number of upcoming clinical studies of site-speci cally prepared ADCs may help clarifying if there is a single conjugation chemistry that will become of widespread use, or whether other methods will also be applicable. Therefore, developing various technol- ogies is of interest for further progress in the eld. Site-speci c conjugation to an antibody is challenging due to the large number of solvent-exposed nucleophilic amino acids, in particular lysines. Despite this di ffi culty, the eld has been very proli c through developing a wide array of technologies that can be summarized as engineered cysteines, disul de
Site-selective template-directed synthesis of antibody Fc conjugates with concomitant ligand release
主要关键词