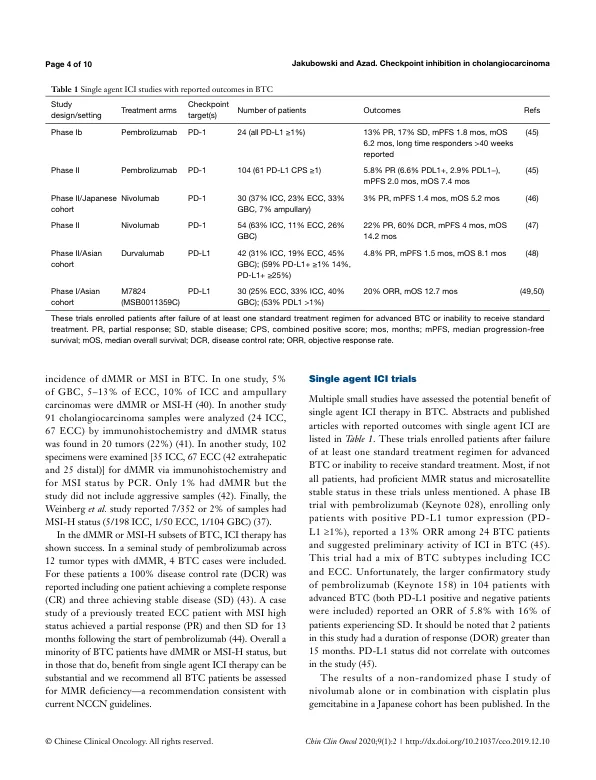
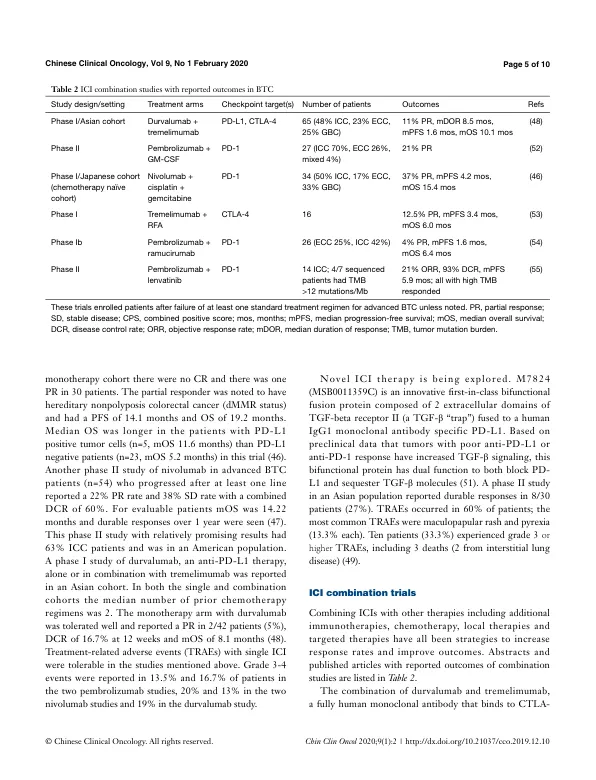
5.0 months and mOS 8.1 months in gemcitabine alone arm) (9). There is no established second line treatment for advanced BTC, but a recent randomized phase III trial (ABC-06) showed a mOS for 5-fluorouracil and oxaliplatin (FOLFOX) of 6.2 months in patients with disease progression after gemcitabine plus cisplatin (10). These outcomes will form the basis of comparison to the trials described in this review. With limited treatment options and dismal overall prognosis, new therapies and approaches are an urgent unmet need in BTC (11). Clinical trials using targeted agents have historically shown only modest responses in BTC patients, though there has been recent success in treating cholangiocarcinomas with isocitrate dehydrogenase-1 (IDH1) and FGFR genetic alterations with novel targeted agents (12-15). Even in these settings, responses have variable durability and more effective, durable therapies are needed. Immunotherapy has had tremendous success in treating patients with cancers such as melanoma and non-small cell lung cancer, dramatically altering the natural history of these diseases. In BTC, immunotherapy strategies have included autologous cell transfer, vaccinations and immunomodulatory approaches with immune checkpoint inhibitors (ICIs), the latter of which is the largest area of ongoing research. In this review, we describe the rationale for ICI therapy by summarizing the clinically relevant tumor immunology in BTC including the innate and adaptive immune response and immune checkpoint molecule expression in BTC. Subsequently we discuss completed and ongoing clinical trials involving ICI in BTC.
胆道癌的免疫检查点抑制剂治疗(...